Researchers identify possible role of Foxp1 protein in control of autoimmune diseases
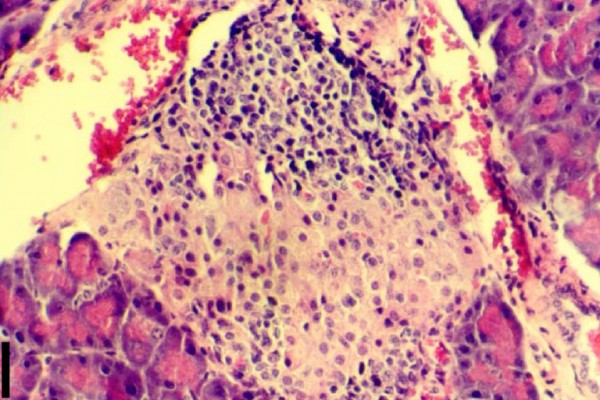
Deutsch: Histologisches Bild einer Insulitis, einer entzündlichen Infiltration der Langerhans-Inseln in der Bauchspeicheldrüse, die der Entstehung eines manifesten Autoimmun-Diabetes vorausgeht. Das Bild basiert auf einer Hämatoxylin-Eosin-Färbung von paraffin-eingebetteten Pankreas-Schnitten transgener PEPCK-Ins NOD-Mäuse. Der schwarze Balken in der linken unteren Ecke repräsentiert eine Strecke von 20µm. Das nach Wikimedia Commons hochgeladene Bild ist eine beschnittene Version des Originalbildes, um eine in der veroffentlichten Version vorhandene Bildbeschriftung zu entfernen.
Scientists at the Higher School of Economics, the Institute of Bioorganic Chemistry of the Russian Academy of Sciences (IBCh RAS), and the Memorial Sloan Kettering Cancer Center created a genetic model that helps to understand how the body restrains autoimmune and oncological diseases. The researchers published their results in Nature Immunology.
The immune systems of humans and animals enable them to resist infectious diseases. That is, they recognize pathogens such as viruses, bacteria, fungi, protozoa, and multicellular parasites and destroy them. The T lymphocyte or "helper lymphocyte" is a special type of immune cell that identifies pathogens and helps other immune cells destroy both the pathogens and the cells they infect. The helper lymphocytes also contain a specialized lineage, called a T-regulatory or "Treg" that, instead of helping to fight infection, actually inhibits the response of normal lymphocytes. Mutations that interfere with the development and proper functioning of Treg cells lead to disastrous consequences for the body. Mice and humans without Treg cells develop fatal autoimmune diseases caused by T-helper cell's uncontrolled attack on the body's own cells.
The scientists studied the properties of the Foxp3 protein that is responsible for the development and proper functioning of Treg cells. They found that removing the Foxp3 protein gene from the genomic DNA prevents the development of Treg cells, leading to the death of the organism. It is also known that numerous autoimmune diseases are associated with abnormal Foxp3 synthesis and Treg cell quantities. The Foxp3 protein does not work alone but as part of a complex of proteins that help it regulate the work of genes necessary for the proper functioning of Treg cells. That set of proteins includes Foxp1, which has been the subject of much less research.
The authors of this study, under the guidance of Aleksander Rudensky, created a genetic model to explain exactly how the Foxp1 protein affects Foxp3. They began by removing part of the Foxp1 gene in Treg from laboratory mice. A comparison of the "normal" cells with the cells in which Foxp1 had been removed revealed that Foxp3 is much worse at binding DNA in the absence of Foxp1. That is, the genes of the proteins crucial for the proper functioning of Treg cells do not work correctly without Foxp1. Thus, if Foxp3 is essential for Treg, then Foxp1 also holds great importance because its removal negatively affects Foxp3. According to the researchers, achieving an understanding of the structure of the complex of proteins that includes Foxp3 and Foxp1 is the key to creating drugs that can selectively affect Treg cells.
'The results significantly broaden our knowledge of the molecular mechanisms regulating immunological tolerance that can be used for treating cancer and autoimmune diseases,' notes Yury Rubtsov, a co-author of the study, associate professor of the HSE faculty of Biology and Biotechnologies, and senior researcher at the IBCh RAS Laboratory of Molecular Oncology. 'Cancerous tumours attract Treg cells to defend themselves against the body's immune system. The more Treg cells present in the tumour, the worse the patient's prognosis. Thus, if we could control the quantity and activity of Treg cells by, for example, decreasing them in the case of a tumour or, on the contrary, increasing them in the case of autoimmune disease, we could create safe medicines for treating heretofore incurable illnesses.'
Feb 25, 2019 11:27 AM EST